I. Mejora de la precisión sintética y la fiabilidad de los datos
En la construcción de una molécula PROTAC, el enlazador no es inerte; sus sutiles cambios impactan directamente en el rendimiento de la molécula final. El PEG polidisperso tradicional es una mezcla de moléculas con diferentes grados de polimerización. Su uso como enlazador da como resultado la síntesis de un PROTAC que, en efecto, también es una mezcla. Esto conlleva dos problemas graves: primero, la variación entre lotes en la distribución de longitudes de polímero dificulta la reproducción de los datos farmacológicos; segundo, durante el cribado y la optimización, los cambios observados en la actividad no pueden atribuirse de forma fiable a cambios en la longitud del enlazador, sino a la interferencia de otros componentes de la mezcla.
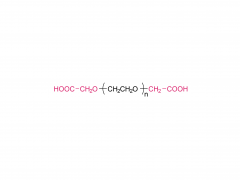
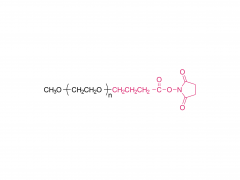
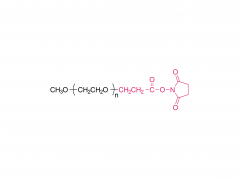
PEG monodisperso
Este método resuelve fundamentalmente este problema. Compuesto por moléculas con un peso molecular único y definido, garantiza que cada molécula de PROTAC sintetizada tenga una estructura y longitud de cadena idénticas. Esta homogeneidad química absoluta es la base para establecer relaciones estructura-actividad (SAR) fiables. Los investigadores pueden tener la certeza de que un aumento o disminución de la actividad refleja directamente la contribución de esa longitud de enlace específica, lo que proporciona datos experimentales auténticos y reproducibles que guían claramente los esfuerzos posteriores de optimización del fármaco.
II. Optimización de las propiedades fisicoquímicas y farmacológicas
Las moléculas PROTAC suelen estar formadas por dos ligandos hidrofóbicos unidos por un enlace, lo que a menudo resulta en una baja solubilidad en agua y una tendencia a agregarse o precipitar en experimentos biológicos. Las cadenas monodispersas de PEG poseen buena hidrofilicidad y flexibilidad. Su incorporación como enlaces puede mejorar eficazmente las propiedades fisicoquímicas generales del PROTAC. Al actuar como una "cuerda hidrofílica", ayudan a solubilizar las porciones de ligando hidrofóbico, aumentando la solubilidad acuosa del PROTAC y previniendo su agregación en condiciones fisiológicas.
Además, la introducción de una cadena de PEG puede modular el comportamiento farmacocinético del PROTAC in vivo. Al aumentar el peso molecular total, reduce la tasa de filtración renal, prolongando así la semivida del fármaco en el torrente sanguíneo. Esto proporciona más tiempo para que el fármaco alcance su tejido diana y ejerza su efecto, lo cual es fundamental para mejorar la eficacia in vivo de los PROTAC y optimizar las pautas de dosificación.
III. Mejora significativa de la actividad biológica
El mecanismo de acción de los PROTAC se basa en su unión simultánea a la proteína diana y a una ubiquitina ligasa E3, formando un complejo ternario estable. La eficacia de este proceso depende en gran medida de si la longitud y la geometría del enlazador permiten que ambos componentes funcionen de forma óptima. En este caso, el enlazador de PEG monodisperso actúa como un regulador de precisión.
Ajustando con precisión la longitud de la cadena de PEG (por ejemplo, PEG2, PEG4, PEG6, etc.), se puede regular con exactitud la distancia espacial y la orientación relativa entre los dos ligandos para encontrar la geometría óptima que permita la formación de un complejo ternario estable. El impacto de esta optimización es significativo: numerosos estudios han confirmado que la introducción de un enlazador de PEG monodisperso adecuado puede aumentar la potencia de degradación de un PROTAC (normalmente medida por la concentración de degradación media máxima, DC50) varias veces o incluso diez veces, alcanzando tasas de degradación máximas (Dmax) superiores al 90 %. Esto demuestra que un enlazador de PEG bien diseñado puede transformar una molécula moderadamente activa en un degradador de proteínas potente y altamente eficaz.
IV. Acelerar el proceso de selección y desarrollo de fármacos.
En las primeras etapas del descubrimiento de fármacos, explorar rápidamente el espacio químico e identificar las moléculas óptimas es crucial. Los enlazadores PEG monodispersos, con su estructura definida y sitios de reacción fijos, se adaptan perfectamente a estrategias de síntesis modulares de alto rendimiento. Los investigadores pueden combinar rápidamente diferentes ligandos de proteínas diana, fragmentos PEG monodispersos de distinta longitud y diferentes ligandos de ligasa E3, como si se tratara de un juego de construcción. Este modelo "plug-and-play" permite la creación de una biblioteca de compuestos con cientos de moléculas PROTAC en muy poco tiempo (por ejemplo, de un día para otro), acelerando drásticamente el descubrimiento y la optimización de compuestos principales.
Las ventajas del PEG monodisperso se mantienen cuando una molécula candidata avanza desde la fase de cribado inicial hasta los estudios toxicológicos posteriores y la ampliación del proceso. Dado que la ruta sintética y los métodos analíticos son consistentes desde la síntesis a escala de miligramos hasta la de gramos, la transferencia de tecnología durante el desarrollo se realiza sin problemas, lo que reduce significativamente el tiempo transcurrido desde el descubrimiento en el laboratorio hasta el desarrollo preclínico y mejora la eficiencia de todo el proceso de investigación y desarrollo.