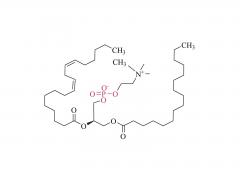
Tras la concesión de la patente europea para lípidos catiónicos en febrero de 2026, nuestra cartera de patentes alcanzó otro hito. En abril de 2026, varios productos de lípidos catiónicos desarrollados de forma independiente por Sinopeg —entre ellos DHA-1, SNP24-1, SNP25-1 y SNP26-1— obtuvieron la patente estadounidense, protegida mediante una fórmula Markush. De este modo, la serie de lípidos catiónicos DHA-1 cuenta ahora con protección de patente en los tres mercados principales de China, Europa y Estados Unidos, consolidando así una sólida posición global en materia de propiedad intelectual para este excipiente clave de los sistemas de administración de LNP.
Título de la patente: Lípido catiónico, liposoma que contiene lípido catiónico y composición farmacéutica de ácido nucleico que contiene liposoma, así como su formulación y aplicación.
Número de solicitud de patente: 18/269,728
Solicitante: Xiamen Sinopeg Biotech Co., Ltd.
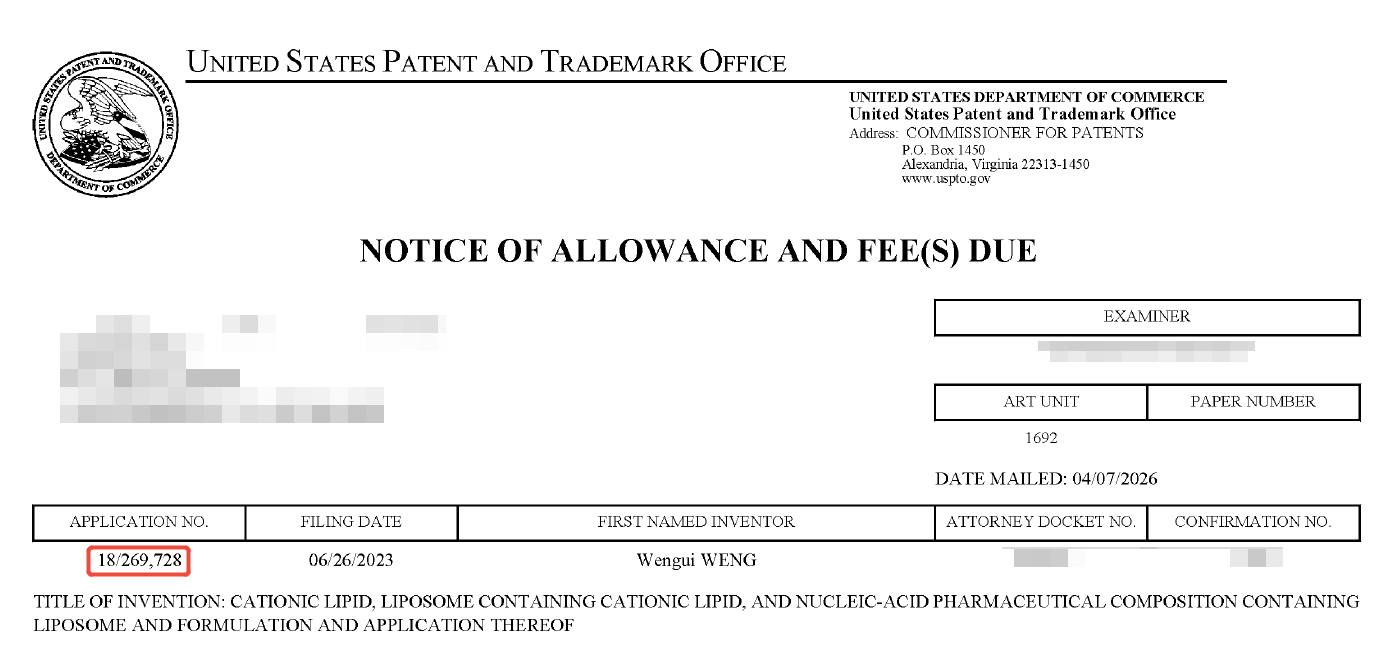
Fuente: Notificación de autorización de la USPTO (captura de pantalla)
Avance en materia de patentes: Cobertura integral desde China hasta Europa y Estados Unidos.
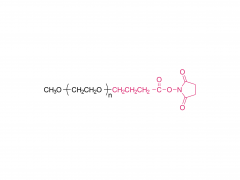
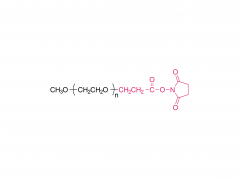
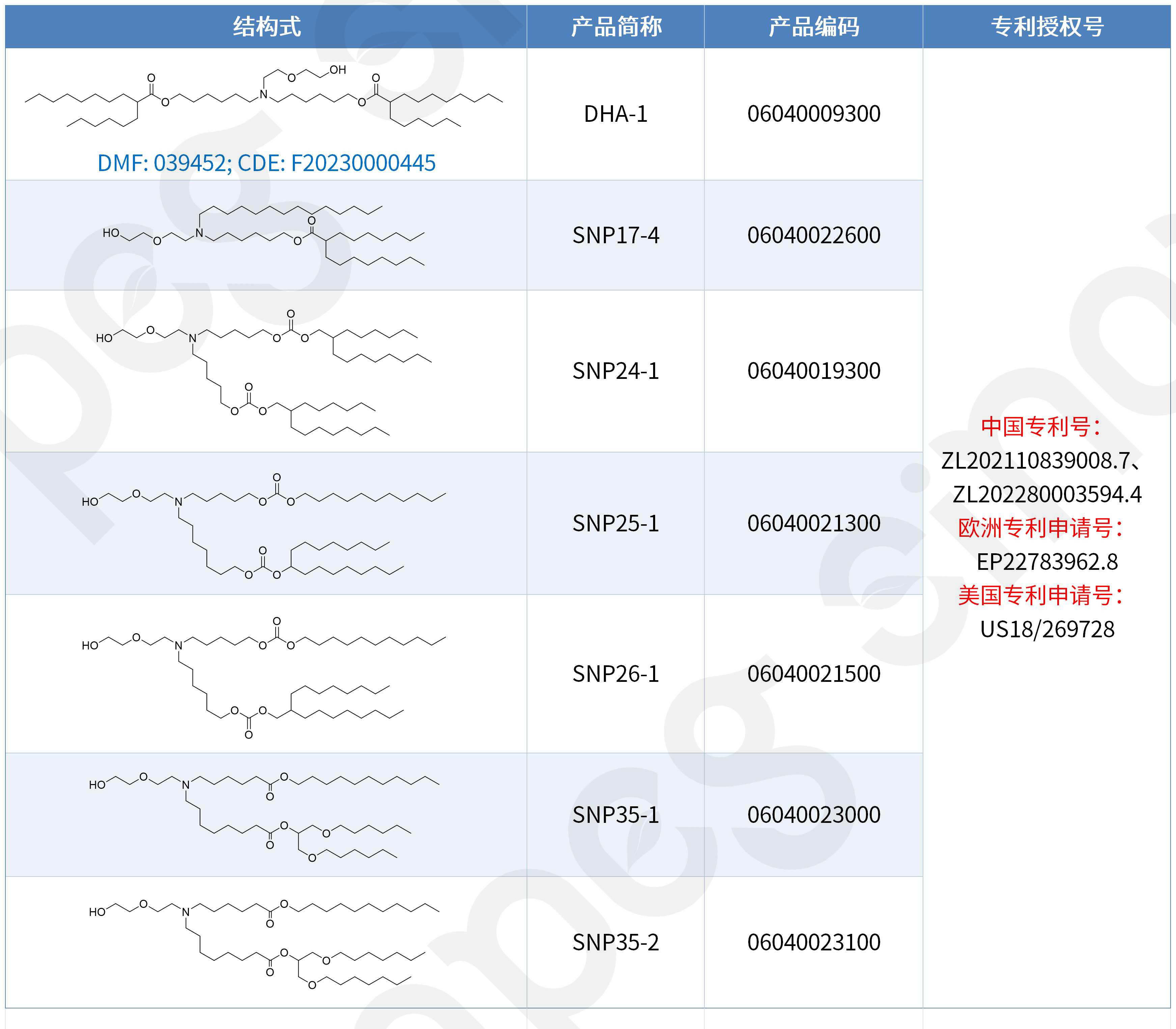
DHA-1, SNP24-1, SNP25-1, SNP26-1 y otros son lípidos catiónicos desarrollados de forma independiente por Sinopeg. Se caracterizan por su alta biocompatibilidad y eficiencia de transfección, lo que permite sortear las restricciones de patentes existentes y ofrecer una alternativa fiable y localizada para la administración de nanopartículas lipídicas.
En lo que respecta a la estrategia de propiedad intelectual, el proceso de obtención de patentes para la serie DHA-1 ha sido sólido y metódico:
Septiembre de 2022: Patente china concedida (solicitud prioritaria).
Octubre de 2023: Se concede la patente para la entrada en la fase nacional del PCT en China.
Febrero de 2026: Se concede la patente europea, logrando una cobertura total en China y Europa.
Abril de 2026: Patente estadounidense concedida, protegida en forma de fórmula Markush.
De este modo, los lípidos catiónicos de la serie DHA-1 cuentan ahora con protección de patente en los tres principales mercados: China, Europa y Estados Unidos, lo que proporciona una sólida base legal para una comercialización conforme a la normativa y una expansión global del negocio. Además de la protección de patente, DHA-1 también destaca por su cumplimiento normativo: ha completado con éxito la presentación de la solicitud de registro como excipiente farmacéutico ante la CDE en China (número de solicitud F20230000445) y ha presentado el DMF ante la FDA de Estados Unidos (número de solicitud 039452). Los clientes pueden consultar directamente los expedientes presentados por Sinopeg tanto en China como en Estados Unidos, lo que reduce significativamente el tiempo de preparación de la solicitud y acelera el progreso del proyecto.
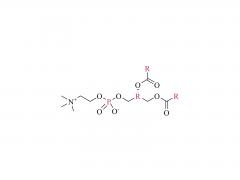
Asignación de la fórmula Markush: Mayor protección, barrera más fuerte
¿Qué es una autorización de fórmula Markush y en qué se diferencia de una patente de compuesto estándar?
En pocas palabras, la fórmula Markush es una de las formas más valiosas de protección de patentes en el campo de los compuestos farmacéuticos. Utiliza un enfoque de "fórmula genérica", que abarca una clase de compuestos que comparten una estructura común y propiedades similares, en lugar de proteger únicamente una sola estructura.
Para esta serie de lípidos catiónicos, esto significa:
Mayor protección: la patente cubre no solo las docenas de estructuras específicas ejemplificadas (incluidas DHA-1, SNP24-1, SNP25-1, SNP26-1), sino también otras estructuras lipídicas catiónicas que comparten el mismo andamiaje central, lo que crea una barrera de patente más sólida.
Es más difícil diseñar soluciones alternativas: los competidores no pueden sortear fácilmente la protección Markush realizando pequeños ajustes estructurales.
Tranquilidad para los clientes: elegir un producto lipídico catiónico protegido por una fórmula Markush ofrece una mayor seguridad para su comercialización.
En resumen, la excepción de la fórmula Markush es una herramienta poderosa para la protección de patentes de compuestos.
Más allá de las patentes estructurales: protección de la propiedad intelectual a nivel del sistema de entrega.
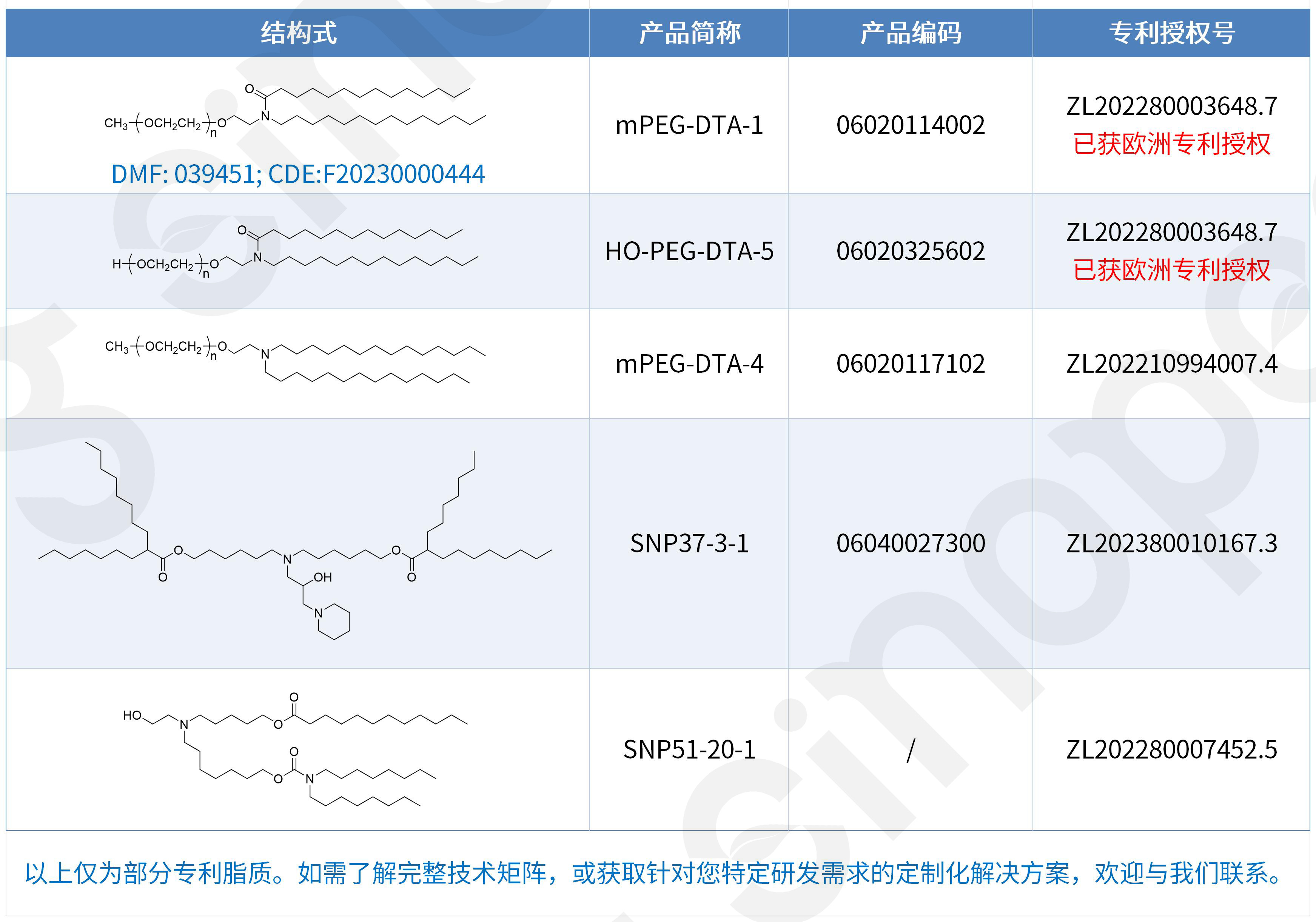
Las patentes de estos lípidos catiónicos en China, Europa y Estados Unidos no solo protegen los compuestos en sí. Las solicitudes de patente de Sinopeg abarcan una solución técnica integral, que va desde el compuesto hasta la composición, incluyendo liposomas o nanopartículas lipídicas, la composición del fármaco de ácido nucleico, la formulación y la aplicación. Se trata de una cartera completa de patentes para sistemas de administración de LNP.
¿Qué significa esto para los clientes?
Si utiliza nuestros productos lipídicos catiónicos en I+D o fabricación, esta protección multicapa —que abarca la materia prima lipídica, la composición lipídica y el sistema de administración— proporciona una seguridad integral de la propiedad intelectual durante todo el ciclo de vida, desde el desarrollo hasta la comercialización, lo que le brinda mayor confianza en sus proyectos.
Además, Sinopeg está buscando activamente carteras de patentes en otros campos de sistemas de administración de fármacos y seguirá profundizando en su experiencia técnica y en la acumulación de propiedad intelectual en soluciones más amplias de sistemas de administración de fármacos (DDS).
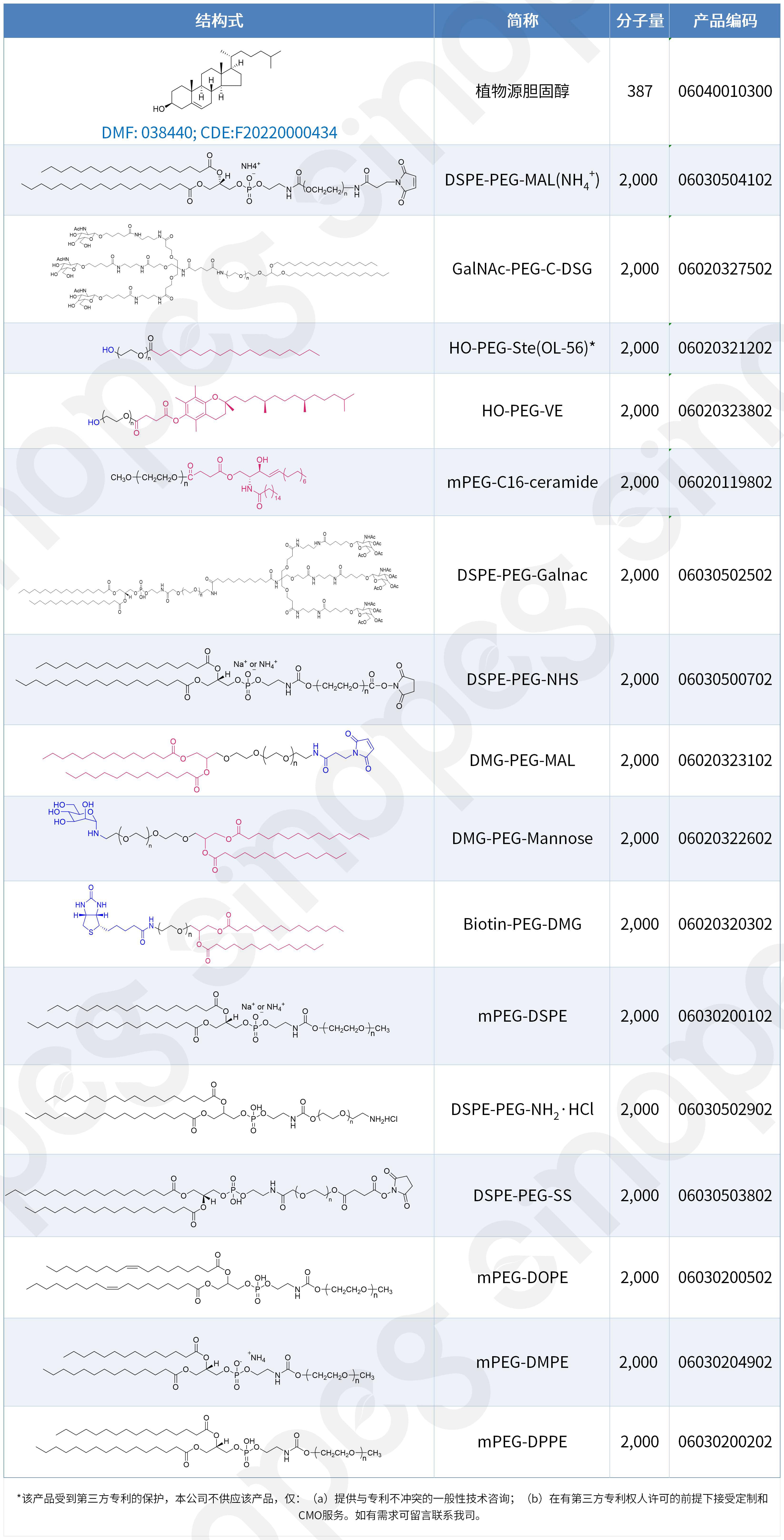
Suministro integral de componentes LNP completos, ampliando nuestra gama de productos.
El suministro estable y la diversidad de componentes son fundamentales para el éxito en la I+D y la fabricación de sistemas de administración de LNP. Sinopeg comprende profundamente las necesidades de sus clientes y ha logrado una capacidad integral para suministrar todos los componentes de los excipientes de los sistemas de administración de LNP. Asimismo, Sinopeg ofrece una amplia gama de productos lipídicos LNP con patentes chinas y europeas, satisfaciendo plenamente las diversas necesidades de los clientes en las etapas de I+D, clínica y producción comercial, a la vez que mitiga eficazmente los riesgos de propiedad intelectual y acelera los plazos de los proyectos.
Conclusión
Desde la concesión de patentes en China hasta la concesión de patentes en Europa, y ahora hasta la aprobación de la fórmula Markush en EE. UU., la trayectoria global de patentes de los lípidos catiónicos de la serie DHA-1 es una prueba del compromiso de Sinopeg con la I+D independiente y su profunda dedicación al campo de los sistemas de administración.
Elegir Sinopeg significa elegir no solo un producto, sino un sistema completo de propiedad intelectual y soporte normativo.
— Para obtener más información sobre la serie DHA-1 y otros productos lipídicos LNP, no dude en ponerse en contacto con nosotros para obtener materiales detallados.